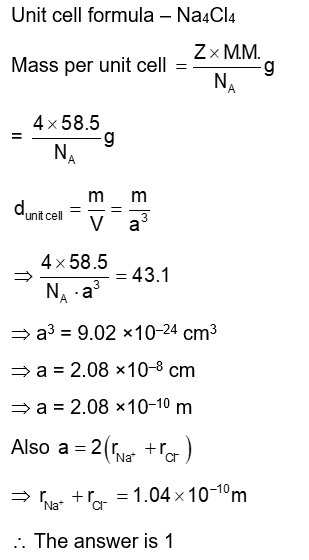The distance between $\mathrm{Na}^{+}$and $\mathrm{Cl}^{-}$ions in 'solid NaCl of density $43.1 \mathrm{~g} \mathrm{~cm}^{-3}$ is $\_\_\_\_$ $\times 10^{-10} \mathrm{~m}$. (Nearest Integer)
(Given : $\mathrm{N}_{\mathrm{A}}=6.02 \times 10^{23} \mathrm{~mol}^{-1}$ )