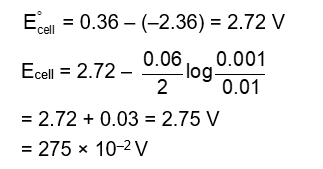The electrode potential of the following half cell at 298 K . $\mathrm{X}\left|\mathrm{X}^{2+}(0.001 \mathrm{M})\right|\left|\mathrm{Y}^{2+}(0.01 \mathrm{M})\right| \mathrm{Y}$ is $\_\_\_\_$ $\times 10^{-2} \mathrm{~V}$ (Nearest integer).
Given:
$$
\mathrm{E}_{\mathrm{x}^2 \mid \mathrm{x}}^0=-2.36 \mathrm{~V}, \mathrm{E}_{\mathrm{Y}^2 \mid \mathrm{Y}}^0=+0.36 \mathrm{~V}, \frac{2.303 \mathrm{RT}}{\mathrm{~F}}=0.06 \mathrm{~V}
$$