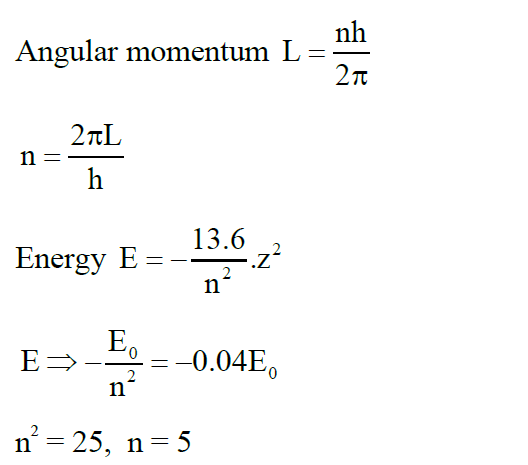The energy of an electron in an orbit of the Bohr's atom is $-0.04 E_0 \mathrm{eV}$ where $\mathrm{E}_0$ is the ground state energy. If $L$ is the angular momentum of the electron in this orbit and $h$ is the Planck's constant, then $\frac{2 \pi L}{h}$ is $\_\_\_\_$ :