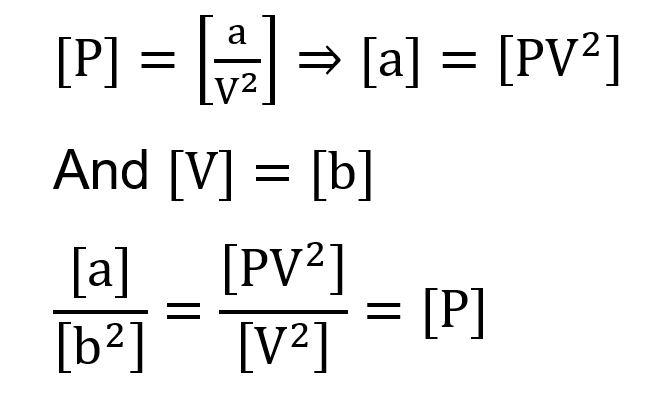The equation of state of a real gas is given by $\left(P+\frac{a}{V^2}\right)(V-b)=R T$, where $P, V$ and $T$ are pressure. volume and temperature respectively and R is the universal gas constant. The dimensions of $\frac{\mathrm{a}}{\mathrm{b}^2}$ is similar to that of: