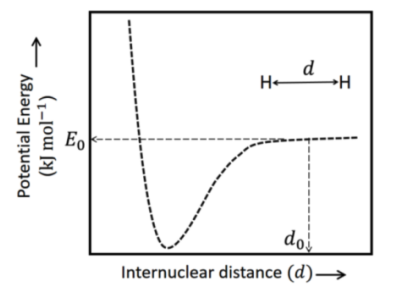
The figure below is the plot of potential energy versus internuclear distance $(d)$ of $\mathrm{H}_2$ molecule in the electronic ground state. What is the value of the net potential energy $E_0$ (as indicated in the figure) in $\mathrm{kJ} \mathrm{mol}^{-1}$, for $d=d_0$ at which the electron-electron repulsion and the nucleus-nucleus repulsion energies are absent? As reference, the potential energy of H atom is taken as zero when its electron and the nucleus are infinitely far apart.
Use Avogadro constant as $6.023 \times 10^{23} \mathrm{~mol}^{-1}$.