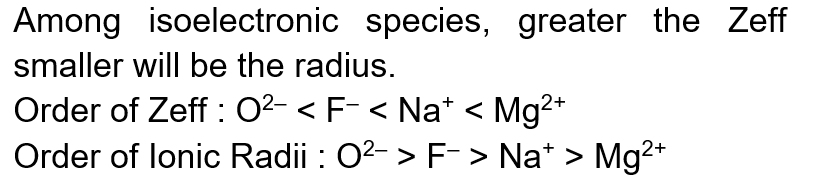Suppose four distinct positive numbers $\mathrm{a}_1, \mathrm{a}_2, \mathrm{a}_3, \mathrm{a}_4$ are in G.P. Let $\mathrm{b}_1=\mathrm{a}_1, \mathrm{~b}_2=\mathrm{b}_1+\mathrm{a}_2, \mathrm{~b}_3=\mathrm{b}_2+ \mathrm{a}_3$ and $\mathrm{b}_4=\mathrm{b}_3+\mathrm{a}_4$. STATEMENT-1:...
JEE AdvanceMathematicsEasy
View Solution→