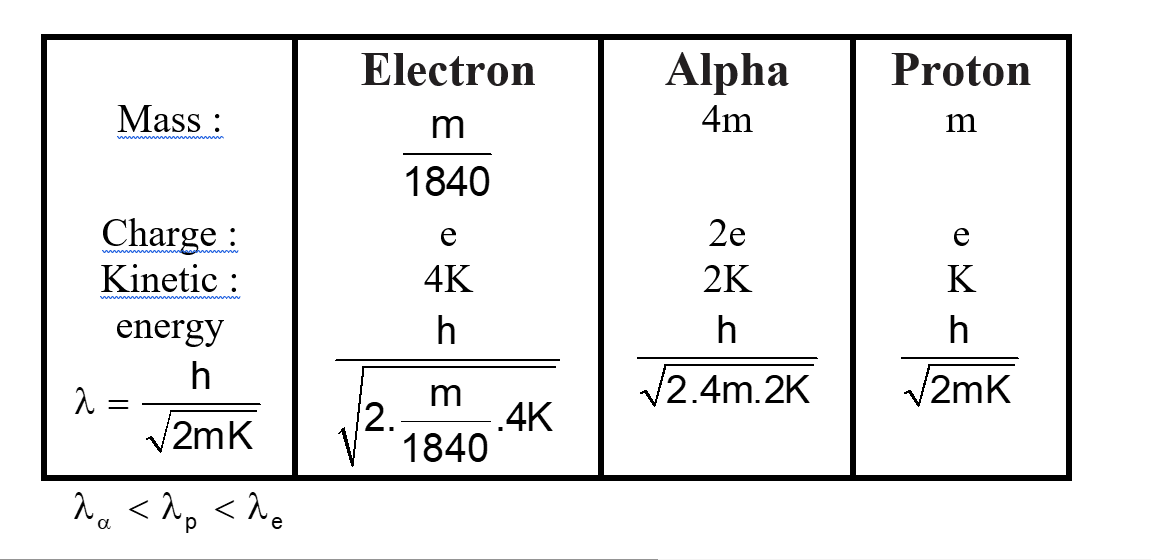
The kinetic energy of an electron, $\alpha$-particle and a proton are given as 4K, 2K and K respectively. The de-Broglie wavelength associated with electron ( $\lambda \mathrm{e}$ ) $\alpha$-particle ( $\lambda \alpha$ ) and the proton ( $\lambda \mathrm{p}$ ) are as follows .