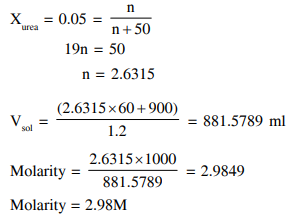The mole fraction of urea in an aqueous urea solution containing 900 g of water is 0.05 . If the density of the solution is $1.2 \mathrm{~g} \mathrm{~cm}^{-3}$, the molarity of urea solution is $\_\_\_\_$
(Given data : Molar masses of urea and water are $60 \mathrm{~g} \mathrm{~mol}^{-1}$ and $18 \mathrm{~g} \mathrm{~mol}^{-1}$, respectively)