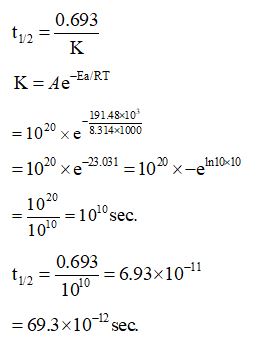${\rm{A}} \to \;{\rm{B}}$
The molecule A changes into its isomeric form B by following a first order kinetics at a temperature of 1000 K. If the energy barrier with respect to reactant energy for such isomeric transformation is $191.48kjmol^{-1}$ and the frequency factor is $10^{20}$ , the time required for molecules of A to become B is _______ picoseconds (nearest integer).$\left[ {{\rm{R}} = 8.314\;{\rm{J}}\;{{\rm{K}}^{ - 1}}\;{\rm{mo}}{{\rm{l}}^{ - 1}}} \right]$