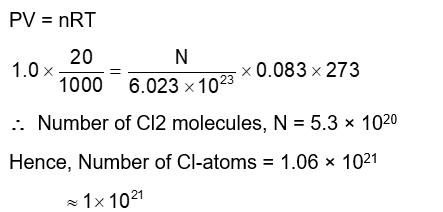
The number of chlorine atoms in 20 mL of chlorine gas at STP is_____$10^2$. (Round off to the Nearest Integer).
[Assume chlorine is an ideal gas at STP
$\left.\mathrm{R}=0.083 \mathrm{~L} \mathrm{bar} \mathrm{mol}^{-1} \mathrm{~K}^{-1}, \mathrm{NA}=6.023 \times 10^{23}\right]$