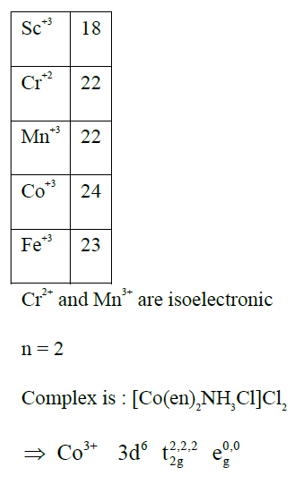
The number of isoelectronic species among $\mathrm{Sc}^{3+}, \mathrm{Cr}^{2+}, \mathrm{Mn}^{3+}, \mathrm{Co}^{3+}$ and $\mathrm{Fe}^{3+}$ is ' $n^{\prime}$. If ' $n$ ' moles of AgCl is formed during the reaction of complex with formula $\mathrm{CoCl}_3(\mathrm{en})_2 \mathrm{NH}_3$ with excess of $\mathrm{AgNO}_3$ solution, then the number of electrons present in the $t_{2 g}$ orbital of the complex is $\_\_\_\_$ .