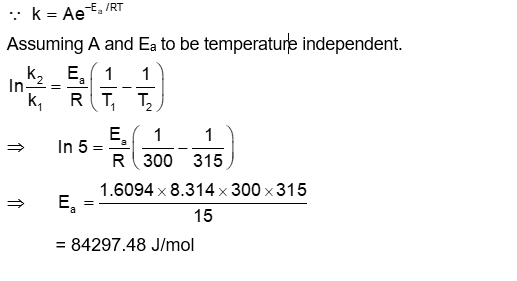
The number of molecules with energy greater than the threshold energy for a reaction increases five fold by a rise of temperature from $27^{\circ} \mathrm{C}$ to $42^{\circ} \mathrm{C}$. Its energy of activation in $\mathrm{J} / \mathrm{mol}$ is $\_\_\_\_$ .
(Take $\ln 5=1.6094 ; \mathrm{R}=8.314 \mathrm{~J} \mathrm{~mol}^{-1} \mathrm{~K}^{-1}$ )