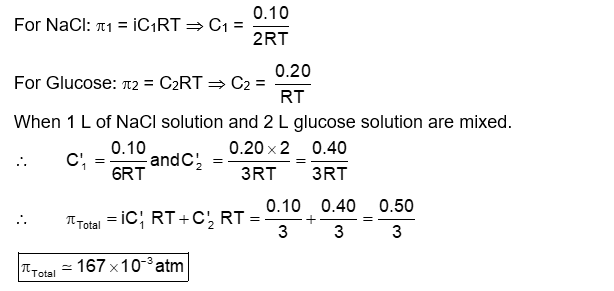
The osmotic pressure of a solution of NaCl is 0.10 atm and that of a glucose solution is 0.20 atm . The osmotic pressure of a solution formed by mixing 1 L of the sodium chloride solution with 2 L of the glucose solution is $\mathrm{x} \times 10^{-3} \mathrm{~atm} . \mathrm{x}$ is $\_\_\_\_$ . (nearest integer)