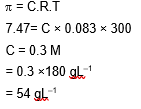The osmotic pressure of blood is 7.47 bar at 300 K . To inject glucose to a patient intravenously, it has to be isotonic with blood. The concentration of glucose solution in $\mathrm{gb}^{-1}$ is $\_\_\_\_$ (Molar mass of glucose $=180 \mathrm{~g} \mathrm{~mol}^{-1}$
$$
\left.\mathrm{R}=0.083 \mathrm{~L} \mathrm{bar} \mathrm{~K}^{-1} \mathrm{~mol}^{-1}\right)
$$
(Nearest integer)