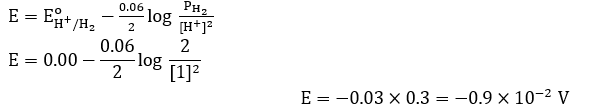The potential for the given half cell at 298 K is $(-) \ldots \ldots \ldots \ldots \times 10^{-2} \mathrm{~V}$.
$2 \mathrm{H}^{+}{ }_{\text {aq }}+2 \mathrm{e}^{-} \rightarrow \mathrm{H}_2(\mathrm{~g})$
$\left[\mathrm{H}^{+}\right]=1 \mathrm{M}, \mathrm{P}_{\mathrm{H}_2}=2 \mathrm{~atm}$
(Given: $2.303 \mathrm{RT} / \mathrm{F}=0.06 \mathrm{~V}, \log 2=0.3$ )