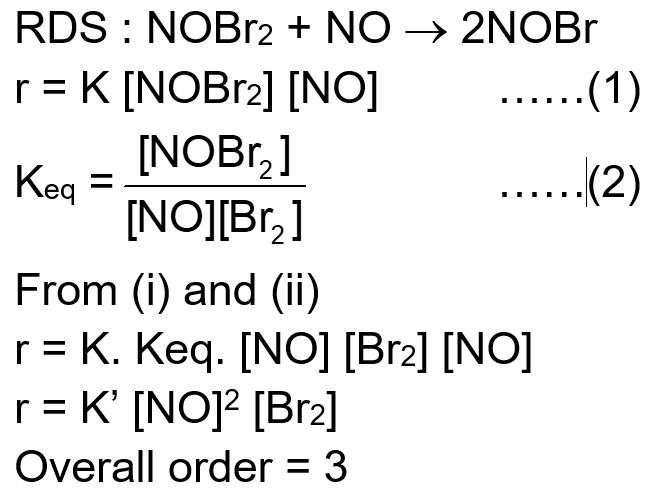
The reaction $2 \mathrm{NO}+\mathrm{Br}_2 \rightarrow 2 \mathrm{NOBr}$ takes places through the mechanism given below:
$\mathrm{NO}+\mathrm{Br}_2 \Leftrightarrow \mathrm{NOBr}_2$ (fast)
$\mathrm{NOBr}_2+\mathrm{NO} \rightarrow 2 \mathrm{NOBr}$ (slow)
The overall order of the reaction is_____.