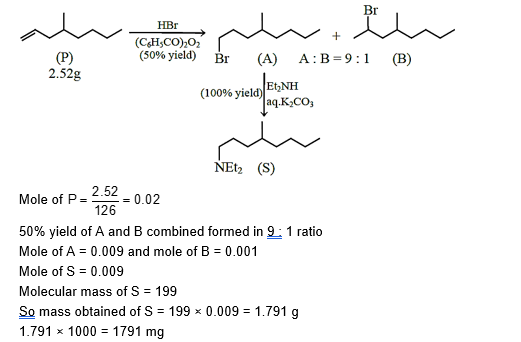
The reaction of 4-methyloct-1-ene $(\mathbf{P}, 2.52 \mathrm{~g})$ with HBr in the presence of $\left(\mathrm{C}_6 \mathrm{H}_5 \mathrm{CO}\right)_2 \mathrm{O}_2$ gives two isomeric bromides in a 9.1 ratio, with a combined yield of $50 \%$. Of these, the entire amount of the primary alkyl bromide was reacted with an appropriate amount of diethylamine followed by treatment with aq. $\mathrm{K}_2 \mathrm{CO}_3$ to give a non-ionic product $S$ in $100 \%$ yield.
The mass (in mg ) of $\mathbf{S}$ obtained is $\_\_\_\_$ .
[Use molar mass (in $\mathrm{g} \mathrm{mol}^{-1}$ ): $\mathrm{H}=1, \mathrm{C}=12, \mathrm{~N}=14, \mathrm{Br}=80$ ]