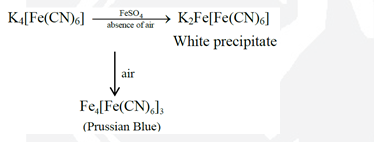
The reaction of $\mathrm{K}_3\left[\mathrm{Fe}(\mathrm{CN})_8\right]$ with freshly prepared $\mathrm{FeSO}_4$ solution produces a dark blue precipitate called Turnbull's blue. Reaction of $\mathrm{K}_4\left[\mathrm{Fe}(\mathrm{CN})_6\right]$ with the $\mathrm{FeSO}_4$ solution in complete absence of air produces a white precipitate X , which turns blue in air. Mixing the $\mathrm{FeSO}_4$ solution with $\mathrm{NaNO}_3$, followed by a slow addition of concentrated $\mathrm{H}_2 \mathrm{SO}_4$ through the side of the test tube produces a brown ring.