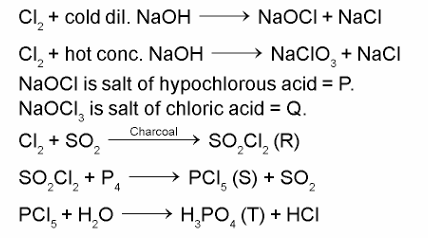
The reactions of $\mathrm{Cl}_2$ gas with cold-dilute and hot-concentrated NaOH in water give sodium salts to two (different) oxoacids of chlorine, $\mathbf{P}$ and $\mathbf{Q}$, respectively. The $\mathrm{Cl}_2$ gas reacts with $\mathrm{SO}_2$ gas, in presence of charcoal, to give a product $\mathbf{R}$. $\mathbf{R}$ reacts with white phosphours to give a compound $\mathbf{S}$. On hydrolysis, $\mathbf{S}$ gives an oxoacid of phosphours $\mathbf{T}$.
P and Q, respectively, are the sodium salts of