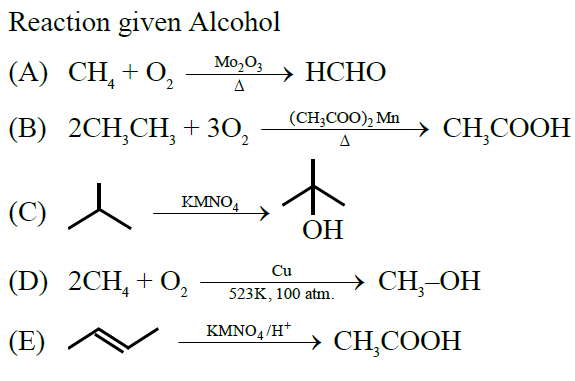
The reactions which produce alcohol as the product are:
A. $\mathrm{CH}_4+\mathrm{O}_2 \xrightarrow[\Delta]{\mathrm{Mo}_2 \mathrm{O}_3}$
B. $2 \mathrm{CH}_3 \mathrm{CH}_3+3 \mathrm{O}_2 \xrightarrow[\Delta]{\left(\mathrm{CH}_3, \mathrm{COO}\right)_2 \mathrm{Mn}}$
C. $\left(\mathrm{CH}_3\right)_3 \mathrm{CH} \xrightarrow{\mathrm{KMnO}_4}$
D. $2 \mathrm{CH}_4+\mathrm{O}_2 \xrightarrow{\mathrm{Cu} / 523 \mathrm{~K} / 100 \mathrm{am} .}$
E. $\mathrm{CH}_3-\mathrm{CH}=\mathrm{CH}-\mathrm{CH}_3 \xrightarrow{\mathrm{KMnO}_4 / \mathrm{H}^{+}}$
Choose the correct answer from the options given below :