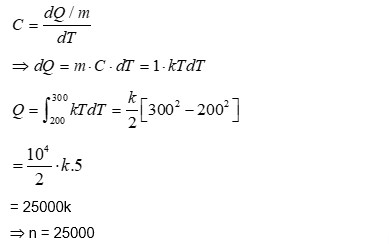The specific heat capacity of a substance is temperature dependent and is given by the formula $C=k T$, where $k$ is a constant of suitable dimensions in SI units, and $T$ is the absolute temperature. If the heat required to raise the temperature of 1 kg of the substance from $-73^{\circ} \mathrm{C}$ to $27^{\circ} \mathrm{C}$ is nk , the value of $n$ is $\_\_\_\_$ .
[Given: $0 \mathrm{~K}=-273^{\circ} \mathrm{C}$.]