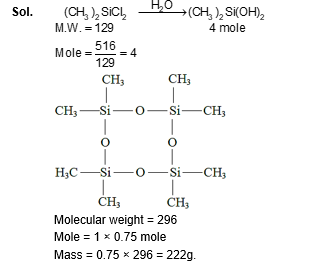
The stoichiometric reaction of 516 g of dimethyldichlorosilane with water results in a tetrameric cyclic product X in $75 \%$ yield. The weight (in g ) of X obtained is $\_\_\_\_$ .
[Use, molar mass $\left(\mathrm{g} \mathrm{mol}^{-1}\right): \mathrm{H}=1, \mathrm{C}=12, \mathrm{O}=16, \mathrm{Si}=28, \mathrm{Cl}=35.5$ ]