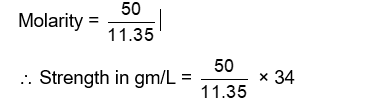The strength of 50 volume solution of hydrogen peroxide is $\_\_\_\_$ $\mathrm{g} / \mathrm{L}$ (Nearest integer).
Given: Molar mass of $\mathrm{H}_2 \mathrm{O}_2$ is $34 \mathrm{~g} \mathrm{~mol}^{-1}$, Molar volume of gas at STP $=22.7 \mathrm{~L}$.