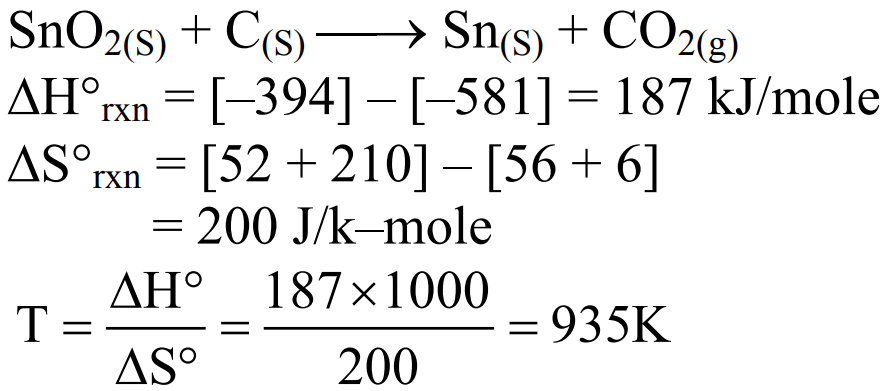Tin is obtained from cassiterite by reduction with coke. Use the data given below to determine the minimum temperature (in K ) at which the reduction of cassiterite by coke would take place.
At $298 \mathrm{~K}: \Delta_{\mathrm{f}} \mathrm{H}^{\circ}\left(\mathrm{SnO}_2(\mathrm{~s})\right)=-581.0 \mathrm{~kJ} \mathrm{~mol}^{-1}, \Delta_{\mathrm{f}} \mathrm{H}^{\circ}\left(\mathrm{CO}_2(\mathrm{~g})\right)=-394.0 \mathrm{~kJ} \mathrm{~mol}^{-1}$
$$
\begin{aligned}
& \mathrm{S}^{\circ}\left(\mathrm{SnO}_2(\mathrm{~s})\right)=56.0 \mathrm{~J} \mathrm{~K}^{-1} \mathrm{~mol}^{-1}, \mathrm{~S}^{\circ}(\mathrm{Sn}(\mathrm{~s}))=52.0 \mathrm{~J} \mathrm{~K}^{-1} \mathrm{~mol}^{-1} \\
& \mathrm{~S}^{\circ}(\mathrm{C}(\mathrm{~s}))=6.0 \mathrm{~J} \mathrm{~K}^{-1} \mathrm{~mol}^{-1}, \mathrm{~S}^{\circ}\left(\mathrm{CO}_2(\mathrm{~g})\right)=210.0 \mathrm{~J} \mathrm{~K}^{-1} \mathrm{~mol}^{-1}
\end{aligned}
$$
Assume that the enthalpies and the entropies are temperature independent.