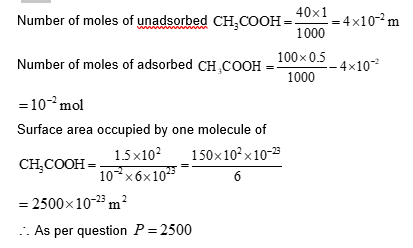To form a complete monolayer of acetic acid on 1 g of charcoal, 100 mL of 0.5 M acetic acid was used. Some of the acetic acid remained unadsorbed. To neutralize the unadsorbed acetic acid, 40 mL of 1 MNaOH solution was required. If each molecule of acetic acid occupies $P \times 10^{-23} \mathrm{~m}^2$ surface area on charcoal, the value of $P$ is $\_\_\_\_$
[Use given data: Surface area of charcoal $=1.5 \times 10^2 \mathrm{~m}^2 \mathrm{~g}^{-1}$; Avogadro's number $\left(\mathrm{N}_{\mathrm{A}}\right)=6.0 \times 10^{23} \mathrm{~mol}^{-1}$ ]