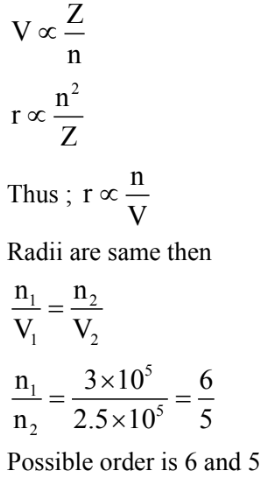Two electrons are moving in orbits of two hydrogen like atoms with speeds $3 \times 10^5 \mathrm{~m} / \mathrm{s}$ and $2.5 \times 10^5 \mathrm{~m} / \mathrm{s}$ respectively. If the radii of these orbits are nearly same then the possible order of energy states are $\_\_\_\_$ respectively.