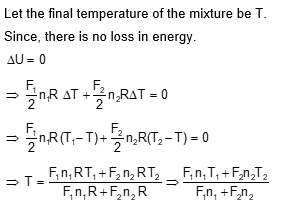Two ideal polyatomic gases at temperatures $\mathrm{T}_1$ and $\mathrm{T}_2$ are mixed so that there is no loss of energy. If $F_1$ and $F_2, m_1$ and $m_2, n_1$ and $n_2$ be the degrees of freedom, masses, number of molecules of the first and second gas respectively, the temperature of mixture of these two gases is: