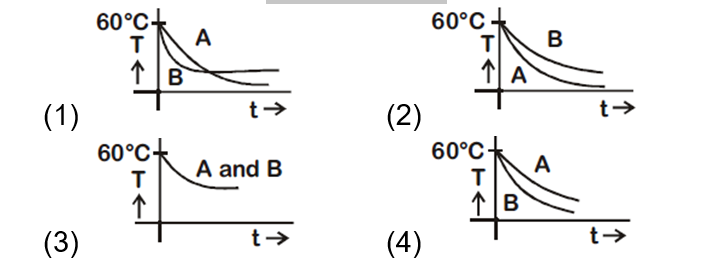
Two identical beakers A and B contain equal volumes of two different liquids at $60^{\circ} \mathrm{C}$ each and left to cool down. Liquid in A has density of $8 \times 10^2 \mathrm{kg} / \mathrm{m}^3$ and specific heat of $2000 \mathrm{Jkg}^{-1} \mathrm{~K}^{-1}$ while liquid in $B$ has density of $103 \mathrm{kgm}-3$ and specific heat of $4000 \mathrm{Jkg}^{-1} \mathrm{~K}^{-1}$. Which of the following best describes their temperature versus time graph schematically? (assume the emissivity of both the beakers to be the same)