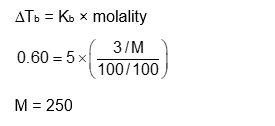
When 3.00 g of a substance ' X ' is dissolved in 100 g of $\mathrm{CCl}_4$, it raises the boiling point by 0.60 K . The molar mass of the substance ' X ' is $\_\_\_\_$ $\mathrm{g} \mathrm{mol}^{-1}$.
(Nearest integer).
[Given $\mathrm{K}_{\mathrm{b}}$ for $\mathrm{CCl}_4$ is $5.0 \mathrm{~K} \mathrm{~kg} \mathrm{~mol}^{-1}$ ]