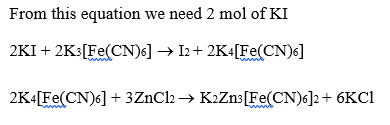When potassium iodide is added to an aqueous solution of potassium ferricyanide, a reversible reaction is observed in which a complex P is formed. In a strong acidic medium, the equilibrium shifts completely towards P. Addition of zinc chloride to P in a slightly acidic medium results in a sparingly soluble complex Q.
The number of zinc ions present in the molecular formula of Q is _____