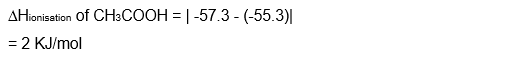While performing a thermodynamics experiment, a student made the following observations,
$
\begin{aligned}
& \mathrm{HCl}+\mathrm{NaOH} \rightarrow \mathrm{NaCl}+\mathrm{H}_2 \mathrm{O} \Delta \mathrm{H}=-57.3 \mathrm{~kJ} \mathrm{~mol}^{-1} \\
& \mathrm{CH}_3 \mathrm{COOH}+\mathrm{NaOH} \rightarrow \mathrm{CH}_3 \mathrm{COONa}+\mathrm{H}_2 \mathrm{O} \\
& \Delta \mathrm{H}=-55.3 \mathrm{~kJ} \mathrm{~mol}^{-1} .
\end{aligned}
$
The enthalpy of ionization of $\mathrm{CH}_3 \mathrm{COOH}$ as calculated by the student is $\_\_\_\_$ $\mathrm{kJ} \mathrm{mol}^{-1}$. (nearest integer)