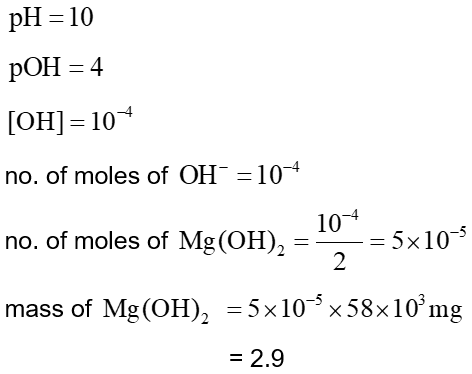$x \mathrm{mg}$ of $\mathrm{Mg}(\mathrm{OH})_2($ molar mass $=58)$ is required to be dissolved in 1.0 L of water to produce a pH of 10.0 at 298 K . The value of $x$ is $\_\_\_\_$ . mg. (Nearest integer)
(Given : $\mathrm{Mg}(\mathrm{OH})_2$ is assumed to dissociate completely in $\mathrm{H}_2 \mathrm{O}$ ]