Using the information which is available in three columns of table given below, give answer of questions Q.31., Q. 32 and Q.33.
Wave function $\psi_{\mathrm{n}, \mathrm{l}, \mathrm{m}_1}$ is a mathematical function, which value depends on spherical polar
coordinates ( $r, \theta, \phi$ ) and titrated from quantum numbers $n$, $I$ and $m_1$. Here, $r$ is distance from nucleus,
$\theta$ is Colatitude and $\phi$ is azimuth. In mathematical functions which are given below table, Z is atomic
number and $\mathrm{a}_0$ is Bohr Radius.
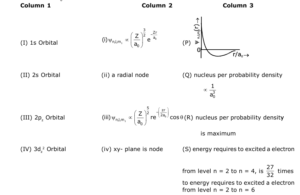
For orbital which are given in column 1, which of the following options has correct combination
for any hydrogen like species :