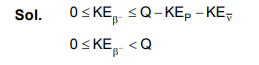The $\beta$-decay process, discovered around 1900, is basically the decay of a neutron (n), In the laboratory, a proton ( p ) and an electron ( $\mathrm{e}^{-}$) are observed as the decay products of the neutron. therefore, considering the decay of a neutron as a tro-body dcay process, it was predicted theoretically that thekinetic energy of the electron should be a constant. But experimentally, it was observed that the electron kinetic energy has a continuous spectrum. Considering a three-body decay process, i.e. $\mathrm{n} \rightarrow \mathrm{p}+\mathrm{e}^{-}+\overline{\mathrm{v}}_{\mathrm{e}}$, around 1930, Pauli explained the observed electron energy spectrum. Assuming the anti-neutrino ( $\bar{v}_e$ ) to be massless and possessing negligible energy, and neutron to be at rest, momentum and energy conservation principles are applied. From this calculation, the maximum kinetic energy of the lectron is $0.8 \times 10^6 \mathrm{eV}$. The kinetic energy carried by the proton is only the recoil energy.
If the anti-neutrino had a mass of $3 \mathrm{eV} / \mathrm{c}^2$ (where c is the speed of light) instead of zero mass, what should be the range of the kinetic energy, K , of the electron?