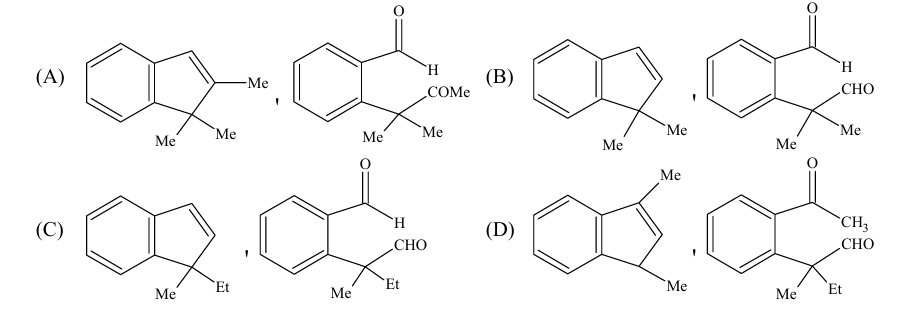
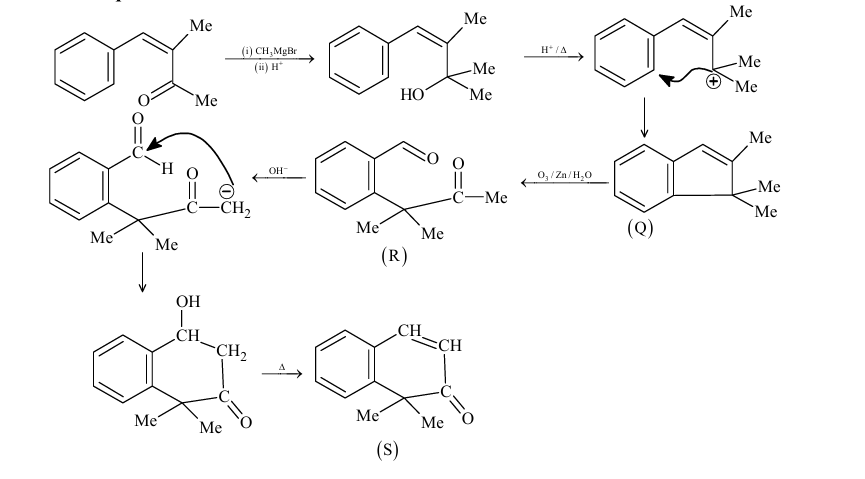
A carbonyl compound $\mathbf{P}$, which gives positive iodoform test, undergoes reaction with MeMgBr followed by dehydration to give an olefin $\mathbf{Q}$. Ozonolysis of $\mathbf{Q}$ leads to a dicarbonyl compound $\mathbf{R}$, which undergoes intramolecular aldol reaction to give predominantly $\mathbf{S}$.
$$
\mathbf{P} \xrightarrow[\substack{2 . \mathrm{H}^{+}, \mathrm{H}_2 \mathrm{O} \\ 3 . \mathrm{H}_2 \mathrm{SO}_4, \Delta}]{\text { 1. } \mathrm{MeMgBr}} \mathbf{Q} \xrightarrow[\text { 2. } \mathrm{Zn}, \mathrm{H}_2 \mathrm{O}]{\text { 1. } \mathrm{O}_3} \mathbf{R} \xrightarrow[\text { 2. } \Delta]{\text { 1. } \mathrm{OH}^{-}} \mathbf{S}
$$
The structure of the products $Q$ and $R$, respectively, are