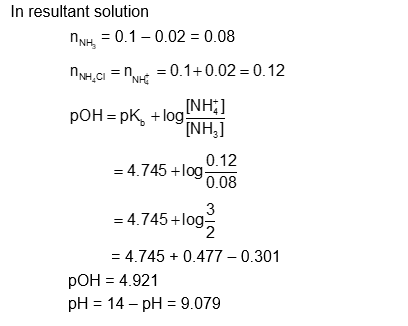
A litre of buffer solution contains 0.1 mole of each of $\mathrm{NH}_3$ and $\mathrm{NH}_4 \mathrm{Cl}$. On the addition of 0.02 mole of HCl by dissolving gaseous HCl , the pH of the solution is found to be $\_\_\_\_$ $\times 10^{-3}$ (Nearest integer)
[Given: $\left.\mathrm{pKb}\left(\mathrm{NH}_3\right)=4.745, \log 2=0.301, \log 3=0.477, \mathrm{~T}=298 \mathrm{~K}\right]$