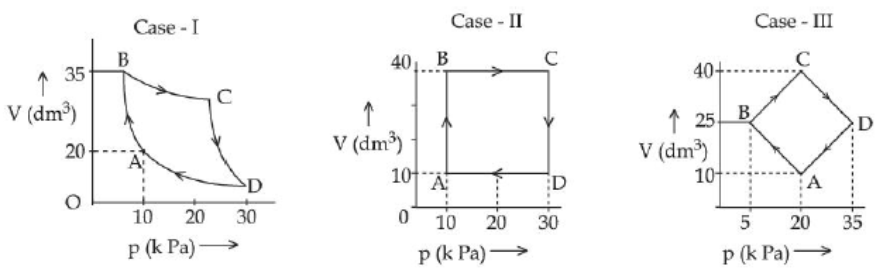
 An ideal gas undergoes a cyclic transformation starting from the point $A$ and coming back to the same point by tracing the path $\mathrm{A} \rightarrow \mathrm{B} \rightarrow \mathrm{C} \rightarrow \mathrm{D} \rightarrow \mathrm{A}$ as shown in the three cases above.
Choose the correct option regarding $\Delta \mathrm{U}$ :
An ideal gas undergoes a cyclic transformation starting from the point $A$ and coming back to the same point by tracing the path $\mathrm{A} \rightarrow \mathrm{B} \rightarrow \mathrm{C} \rightarrow \mathrm{D} \rightarrow \mathrm{A}$ as shown in the three cases above.
Choose the correct option regarding $\Delta \mathrm{U}$ :