Given below are two statements, one is labelled as
Assertion A and the other is labelled as Reason R.
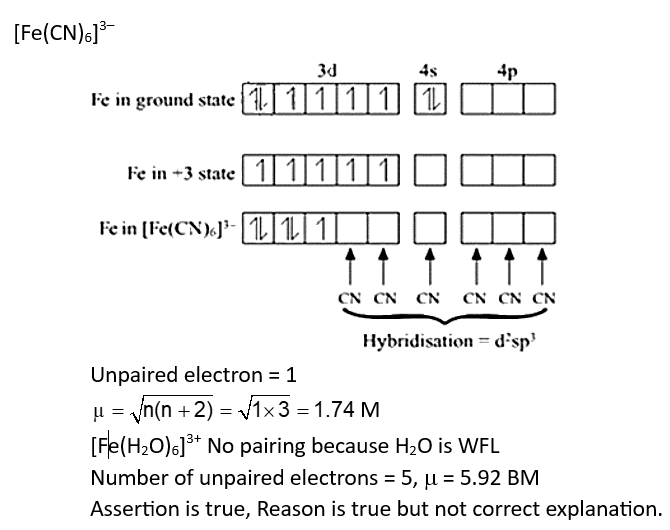
Assertion A: The spin only magnetic moment value for $\left[\mathrm{Fe}(\mathrm{CN})_6\right]^{3-}$ is 1.74 BM , whereas for $\left[\mathrm{Fe}\left(\mathrm{H}_2 \mathrm{O}\right)_6\right]^{3+}$ is 5.92 BM.
Reason R : In both complexes, Fe is present in +3 oxidation state.
In the light of the above statements, choose the correct answer from the options given below: