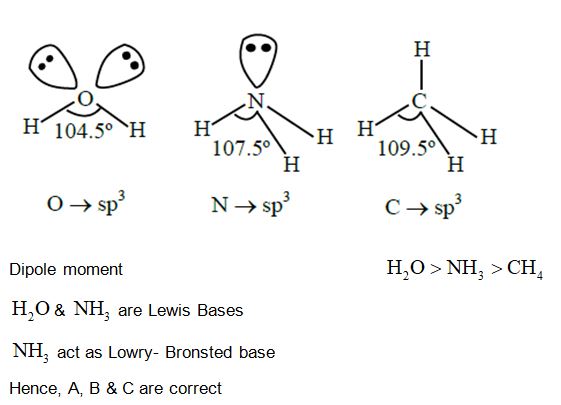
Which of the following statement is true with respect to ${{\rm{H}}_2}{\rm{O}},{\rm{N}}{{\rm{H}}_3}$ and ${\rm{C}}{{\rm{H}}_4}$ ?
A. The central atoms of all the molecules are ${\rm{s}}{{\rm{p}}^3}$ hybridized.
B. The ${\rm{H}} - {\rm{O}} - {\rm{H}},{\rm{H}} - {\rm{N}} - {\rm{H}}$ and ${\rm{H}} - {\rm{C}} - {\rm{H}}$ angles in the above molecules are ${104.5^0},{107.5^0}$ and ${109.5^0}$, respectively.
C. The increasing order of dipole moment is ${\rm{C}}{{\rm{H}}_4} < {\rm{N}}{{\rm{H}}_3} < {{\rm{H}}_2}{\rm{O}}$.
D. Both ${{\rm{H}}_2}{\rm{O}}$ and ${\rm{N}}{{\rm{H}}_3}$ are Lewis acids and ${\rm{C}}{{\rm{H}}_4}$ is a Lewis base.
E. A solution of ${\rm{N}}{{\rm{H}}_3}$ in ${{\rm{H}}_2}{\rm{O}}$ is basic. In this solution ${\rm{N}}{{\rm{H}}_3}$ and ${{\rm{H}}_2}{\rm{O}}$ act as Lowry-Bronsted acid and base respectively.
Choose the correct answer from the options given below: