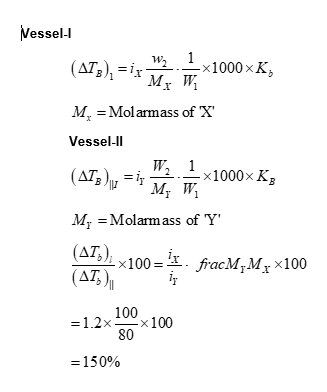Vessel- 1 contains $\mathbf{w}_2 g$ of a non-volatile solute $\mathbf{X}$ dissolved in $\mathbf{w}_1 g$ of water. Vessel- 2 contains $\mathbf{w}_2 g$ of another non-volatile solute Y dissolved in $\mathbf{w}_1 g$ of water. Both the vessels are at the same temperature and pressure. The molar mass of $\mathbf{X}$ is $80 \%$ of that of $\mathbf{Y}$. The van't Hoff factor for $\mathbf{X}$ is 1.2 times of that of $\mathbf{Y}$ for their respective concentrations.
The elevation of boiling point for solution in Vessel-1 is $\_\_\_\_$ \% of the solution in Vessel- 2.